Mirum and Albireo are Interesting Opportunities in Rare Pediatric Liver Diseases
Both of these companies recently got their first approvals and received lucrative rare pediatric disease Priority Review Vouchers. I think both have potential as long-term investments.
Albireo Pharma (ALBO) and Mirum Pharmaceuticals (MIRM) are both developing therapies for rare pediatric liver disorders, and each just received its first FDA approval, with Albireo’s Bylvay being approved for PFIC patients in July and Mirum’s Livmarli receiving approval in ALGS on September 29. Both of these drugs operate through the same mechanism, and both companies trade at very similar market caps. Both companies are intriguing to me, so I’ve spent the last month or so digging into their similarities and differences.
In this article, I’ll go one by one through the initial three potential indications for these companies, briefly review areas of potential expansion beyond the pediatric setting, and take a look at their respective balance sheets and valuations. One initial disclosure though that I want to make before getting started – I have a small, long position in both companies already through cash-secured puts although it is far from what I consider a full position relative to my overall portfolio size.
Also just a note about this newsletter. I recently stepped away from the small subscription service I ran on Seeking Alpha and also stopped publishing articles there. Part of this was due to wanting to take more time for my young family, but part of it was also due to wanting to share my work more openly. There are so many people on Substack and Twitter far more qualified than me that generously share their thoughts on biotech, and I want to return the favor. I will likely publish on a roughly once monthly cadence. This is research I do primarily for my own investing, and writing it up in an article format helps me further process my thoughts. I hope some of you will find the information helpful as well.
Inhibiting Bile Acid Transporters to Treat Pediatric Cholestatic Liver Diseases
All of Mirum and Albireo’s current therapies are intended to address cholestatic liver diseases. These diseases revolve around bile acids, a substance produced from cholesterol by the liver. Bile acids move from the liver into the intestines where they help with the breakdown and absorption of dietary fats and fat-soluble vitamins. Roughly 95% of bile acids in the intestine are then recirculated back into the liver after facilitating this role while the other 5% are excreted in feces. Cholestatic liver diseases are ones where excess bile accumulates within the liver, and this can occur for a variety of reasons depending on the specific disease.
Unfortunately, several varieties of cholestatic liver diseases typically manifest themselves in early childhood and cause at best a very poor quality of life for affected children, and at worst a liver transplant or even death. Oddly one of the most prominent symptoms of these diseases is extreme itching, leading a child to scratch constantly to the point where it can cause substantial harm and even impair sleep and other activities. Excess bile accumulation in the liver can also lead to decreased fat and nutrient intake in the intestines. This is turn causes many children with these diseases to have growth restrictions.
There are no current drug therapies for any of the three pediatric diseases I’m about to discuss other than Livmarli and Bylvay now, so the unmet need is high. Treatments used previously were really primarily just supportive measures intended to help with symptoms like itching and nutrient deficiencies/growth problems and then surgical interventions like a partial biliary diversion or a liver transplant when symptoms became too severe.
Figure 1: Current Treatment for ALGS (source: NORD) (screen capture from video)
This figure was specifically discussing ALGS treatment, but unfortunately the story is pretty similar for PFIC and BA.
Livmarli is an apical sodium-dependent bile acid transporter (ASBT) inhibitor which means that it blocks the recirculation of bile acids back into the liver. Bylvay works on the same pathway, but Albireo refers to these transporters by their alternate name, ileal bile acid transporters (IBAT). By stopping the main way that bile acids are recycled to the liver, Livmarli and Bylvay can decrease bile acid levels in the liver and serum.
Figure 2: Maralixibat’s Mechanism of Action (source: Mirum’s Corporate Presentation)
This mechanism of action is important because both companies have presented data suggesting that lowering serum bile acid levels correlates with improved patient outcomes across these three diseases. This suggests these treatments are potentially modifying the underlying disease process rather than just treating symptoms.
Figure 3: Bile Acid Natural History Study in Biliary Atresia (source: Albireo’s August 2021 Corporate Presentation)
Figure 4: Bile Acid Correlations with Outcomes in PFIC Patients (source: Mirum’s September 2021 Corporate Presentation)
Given that these therapies are the only non-surgical ones that have this potential to truly modify disease progression outcomes, I personally expect to see strong uptake in markets where they end up approved.
The three diseases these companies both chose to address first are progressive familial intrahepatic cholestasis (PFIC), Alagille Syndrome (ALGS), and Biliary Atresia (BA).
PFIC is the rarest of the three main indications discussed here, with likely only about 500 to 750 children in the US needing treatment from a drug like Livmarli or Bylvay at present. The reason PFIC is rarer is because it requires a particular mutation to be inherited from both parents rather than just one like we’ll see with ALGS. Some types of PFIC can occur in infancy while others present in adolescence or later. Problems caused by it can include short stature, deafness, diarrhea, itching, fat-soluble vitamin deficiencies, and even liver failure or liver cancer in severe cases. As the name implies, the bile acid build-up in PFIC patients tends to get worse over time, making many of these worse outcomes more likely as a patient gets older.
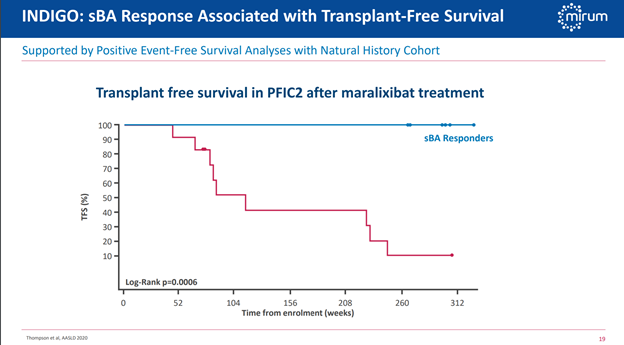
Albireo’s Bylvay is already approved here and for all three subtypes, whereas Livmarli won’t have pivotal data until next year and even then, it may only be for PFIC2 patients. Clear advantage Albireo here but definitely keep in mind that this is the smallest market we’ll be discussing.
Figure 5: The Initial Three Targeted Indications for Both Companies (source: Albireo’s August 2021 Corporate Presentation)
ALGS is caused by one of two different potential genetic mutations that cause bile ducts to be either malformed, excessively narrow, or sometimes even absent entirely. ALGS is a cholestatic liver disease because these bile duct problems cause bile to accumulate in the liver and serum, almost always causing severe itching (pruritis) and sometimes causing other symptoms like xanthomas (accumulations of cholesterol), jaundice, and even liver failure.
ALGS is almost always diagnosed in infants and affects about 2,000 to 2,500 children in the US. Unfortunately, the symptoms associated with ALGS are so severe that only about 25% of all ALGS patients make it to age 18 without having needed a liver transplant.
This is the indication that Livmarli just received its first approval in, while Albireo won’t have its pivotal Phase 3 data until next year. This almost seems like the exact reverse situation as we just saw in PFIC, but again, ALGS is roughly double the potential market opportunity.
BA progresses much more rapidly than ALGS or PFIC and typically requires surgical intervention when an infant is just a few months old. This can be a liver transplant, but it is more commonly something called a Kasai procedure, where a piece of the small intestine is adhered to the liver to create a channel for bile acids to flow out and be excreted. As you might imagine, the surgery is pretty difficult to endure and comes with a substantial physical toll even if successful, but unfortunately, the procedure is only successful about 30% to 40% of the time anyway. Most patients that have a failed Kasai procedure end up then having a liver transplant before even turning two years old.
Albireo expects to have BA data in 2024, and Mirum’s is expected a bit sooner in 2023. I see a high likelihood of success given the strong correlation between bile acid levels and transplant-free survival already known, and transplant-free survival is the primary endpoint here. In fact, the picture I showed in Figure 3 above is from a natural history study of post-Kasai BA patients that shows a clear correlation between serum bile acid levels and patient outcomes. The fact that the endpoint in both companies’ pivotal trials is transplant-free survival here versus the pruritis aspect is what makes them so much longer than the trials for ALGS and PFIC, but the larger market size could make this indication well worth the wait for these companies. Estimates I’ve seen for the number of US patients range from 2,000 to 4,000, so this is another roughly two-fold increase in opportunity size compared to ALGS which itself was around double the PFIC opportunity
Beyond just these three diseases, there are a number of adult cholestatic liver diseases that both companies intend to treat.
Figure 6: Mirum’s Potential Adult Indications (source: Mirum’s September 2021 Corporate Presentation)
Mirum seems to be further ahead in adult indications with potentially pivotal Phase 2b trials underway in multiple indications already. Albireo’s adult-focused therapies are only in Phase 1 right now with data expected later this year.
The main importance of these adult indications is that they are far more common and provide a larger patient population for both companies to eventually expand into.
Overall Points of Differentiation
Beyond time to market, there are a few other things that potentially set these drugs apart from each other.
The most obvious is that one is frequency of use which is once daily for Bylvay (Albireo) versus twice daily for Livmarli (Mirum), but I have a hard time thinking that will change many prescribers’ attitudes in its own. Efficacy or safety would be a much bigger deal, so I looked for whether I saw meaningful differences there. These two drugs appear largely indistinguishable on efficacy to me though. That’s because they both report substantial improvements in the majority of patients in both subjective itch scores and in serum bile acid response, but they don’t ever use the same scale or report exactly the same measure. For example, Mirum tends to report the actual reduction in serum bile acids, while Albireo reports the number of responders seen in its trial. The numbers from both though seem to indicate that these drugs, consistent with their mechanisms, are lowering serum bile acids in the majority of patients which should lead to improved outcomes and symptoms in responders. One drug may in actuality be better than the other, but I’m not sure it’s really possible to tell at this point.
On the whole, these drugs also have a strong safety profile, but diarrhea is one of the few concerns seen that can lead to substantial tolerability problems if severe enough. The rates for both drugs look similar to me at around 40% of patients long-term which is a fairly large proportion of those on treated. That said, none of these cases of diarrhea were reported as serious, and it doesn’t appear that any diarrhea events led to withdrawals so this is almost certainly something patients with a serious liver disease would be willing to tolerate.
Oddly, Albireo’s corporate presentation lists a rate of 9.5% in its PEDFIC 1 trial, but I simply don’t see where they get this number from based on the data reported from their trial.
Figure 7: Adverse Event Reports from the PEDFIC 1 Trial (source: clinicaltrials.gov)
Both companies also plan to commercialize these drugs outside of the US, and both have several partnerships in place already to do so. One potential edge though is that Albireo has one of its main offices in Sweden already and seems to be completely comfortable operating its own European commercialization. This could be an advantage versus Mirum who might have to partner out the opportunity or have a harder time in a less familiar territory.
Albireo is charging $385,000 per year for Bylvay, and Mirum just announced an average annual price of $391,000 for Livmarli. In my modeling, I show a price of $350,000 just to be conservative, but even at that level, sales could be large in comparison to these companies’ market caps. Using Albireo’s market estimates of 1,500 ALGS patients, 600 PFIC, and 2,400 BA, for Albireo, a 40% market share in ALGS would generate $210 million annually while a 60% market share in PFIC would generate $126 million. For Mirum, a 60% market share in ALGS would generate $315 million in peak sales while a 40% market share in PFIC would generate $84 million. Biliary atresia at a 50/50 split for these companies could potentially generate up to another $420 million each in peak sales.
By my estimate, the total addressable market available to these two companies in these three diseases could be close to $1 billion even using Albireo’s market estimates which, for what it’s worth, are generally a bit lower than Mirum’s. That’s plenty of room for both companies to potentially perform really well for shareholders considering that their current combined market caps are not substantially over $1 billion at present.
These Companies’ Balance Sheets and Valuations are Similar
Mirum and Albireo have very similar market caps right now, with roughly a $650 million market cap for Mirum and $625 million market cap for Albireo as of the close on October 1.
Mirum has a pretty large cash balance in comparison to this, with the company reporting $238 million in cash and equivalents at the end of Q2. Since then though, the company just received a priority review voucher that carries a cash value around $100 million, and Mirum also should be receiving $35 million under its deal with Oberland Capital now that Livmarli is approved. Mirum also has no appreciable long-term debt and burned about $44 million in Q2, so I would estimate Mirum’s present enterprise value to be around $325 million currently at the end of Q3. On top of this, Mirum also potentially has another $50 million available to it through the Oberland deal for potential pipeline acquisitions if Mirum and Oberland can agree on such an opportunity.
Albireo similarly doesn’t have significant long-term liabilities aside from a royalty-backed loan, so with $186 million in cash and equivalents at the end of Q2, the $105 million from selling its priority review voucher, and a burn rate around $36 million per quarter, I estimate Albireo’s enterprise value to be around $370 million at the end of Q3.
At this level of enterprise value, I see long-term upside potential in both companies although Mirum seems to be the cheaper of the two right now. Aside from just the price, I personally like Mirum better for three additional reasons. First, Mirum got its product approved in a larger starting market with ALGS rather than PFIC, so not only will it likely build bigger sales quicker, Mirum is likely to hold onto a larger share of that larger market versus Albireo over time due to its first mover advantage and the products’ high degree of similarity.
Figure 8: Sizes of the Initial Three Targeted Markets for Both Companies (source: Albireo’s August 2021 Corporate Presentation)
Second, I really hate seeing companies downplay side effects like it appears Albireo may be doing in presenting its diarrhea events from the PEDFIC 1 trial. This raises a question about management to me that I just don’t have with Mirum right now. Last, while the impact of this is years down the road, I feel like Mirum is at least somewhat ahead in BA and all of the adult cholestatic liver indications. If I had to just chose one of the two companies, that’s a pretty strong reason in favor of Mirum long-term even if both companies were to outperform nearer term.
Conclusion
Both of these companies present a good risk/reward of having successful launches and providing good returns for shareholders. Statistically, drugs like Livmarli and Bylvay that are first-in-class, treat orphan disease, and had priority review are more likely to succeed than the average drug, so I like either company from a long-term standpoint right now, although for the reasons just discussed I favor Mirum.
Figure 9: Relative Odds of Launch Success by Drug Characteristics (source: Deloitte article)
The biggest risk in my mind is that early commercial stage biotechs in general have done really poorly over this past year especially, but I believe there is good reason for these two to potentially be different, although patience may be required to let this thesis play out.
As I said at the beginning, I’ve personally already sold some cash-secured puts in both companies, but they are far less than what I would potentially consider a full position in my current portfolio assuming those shares got assigned. I will almost certainly weight my Mirum position higher relative to any Albireo shares I own over time, and I will likely wait and see what happens with those initial puts and with the Bylvay and Livmarli launches before taking any further action. Specifically, I want to have plenty of dry powder available to build a long-term position at a lower cost basis if the opportunity presents itself like after one of these first few quarterly reports post-launch.
**Please note: I am not a registered investment advisor, and this article is not intended to be investment advice. This is a free newsletter that I publish for entertainment and educational purposes only. You need to do your own diligence based on your own individual situation of which I am not aware.










good work! Love it! Just one remark from my side: Mirum needs to pay milestones plus considerable royalties to shire. Albo does not have to pay this